Retriever Medical pioneers groundbreaking solutions for vascular occlusion with an innovative patent application.
— Brandon Repko, MD
LAS VEGAS, NV, UNITED STATES, March 27, 2024 /EINPresswire.com/ — Retriever Medical, (www.rtvmed.com) a pioneer in interventional medicine, proudly announces a significant milestone in medical innovation with the submission of a U.S. patent application for its groundbreaking Vascular Occlusion Removal System (VORS). Developed by Retriever Medical’s team of experts, the U.S. patent application discloses and claims a revolutionary system and method for removing occlusions and unwanted matter from vascular vessels, promising enhanced outcomes in patient care.
Background of the Invention: The medical field often grapples with challenges in efficiently removing occlusions, such as thrombi, from vessels, particularly in vascular structures like the aorta, pulmonary artery, iliofemoral, and vena cava. Existing devices often struggle with organized embolic material, leading to suboptimal outcomes and potential complications.
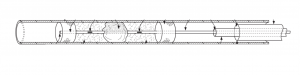
Introducing the Vascular Occlusion Removal System (VORS): Retriever Medical’s VORS represents a significant leap forward in addressing these challenges. The patent application describes a catheter-based system comprising a retriever sub-system with a guide catheter, a delivery catheter, an elongated delivery member, and occlusion-capturing elements. The system provides precise positioning and dynamic adjustment to match the size of the target occlusion, even within the vessel or cavity.
Key features of the VORS system include:
1. Dynamic Surrounding: The retriever sub-system enables dynamic surrounding of the target occlusion, offering superior control and isolation at the
occlusion site.
2. Thrombolytic Agent Delivery: The elongated delivery member of the retriever sub-system includes means for delivering thrombolytic agents, which
enhances the effectiveness of thrombus removal.
3. Pharmacological Agent Delivery: The occlusion-capturing elements of the retriever sub-system are coated with a unique drug-dispersing polymer,
whereby, pharmacological agents, such as anti-inflammatories, can be delivered directly to vessels during an occlusion removal procedure.
According to Brandon Repko, MD, “The submission of this U.S. patent application represents a significant advancement in interventional medicine. The Vascular Occlusion Removal System offers a novel approach to addressing occlusions, providing clinicians with enhanced precision and effectiveness in treatment.”
The Retriever Medical ClotHound ACE thrombectomy systems offer a multitude of advantages over conventional alternatives. With features such as a dual sphere design with distal embolic protection, physician-controlled Clear ACE functionality, single device placement for continuous thrombus removal without the need to remove the device, and integrated mechanical spheres alongside a large bore aspiration catheter, the ClotHound ACE system sets a new standard in thrombectomy technology. Now, with the pending addition of the Vascular Occlusion Removal System (VORS), the ClotHound System is poised to represent the next generation of thrombectomy devices, promising even greater precision, efficiency, and patient outcomes.
Ben Bobo, CEO of Retriever Medical, added, “The submission of our U.S. patent application for the Vascular Occlusion Removal System underscores Retriever Medical’s commitment to driving innovation in interventional medicine. This milestone reinforces our dedication to advancing patient care and improving procedural outcomes.”
About Retriever Medical:
Retriever Medical, Inc. was established with a bold mission to transform interventional medicine by creating groundbreaking surgical solutions, such as the revolutionary ClotHound ACE thrombectomy systems. Demonstrating our dedication to innovation, Retriever Medical has significantly expanded its patent portfolio, comprising eight (7) issued U.S. and two (2) international patents, alongside ten (10) pending U.S. patent applications and an additional ten (10) pending foreign patent applications spanning multiple jurisdictions, including the European Community, Hong Kong, Canada, Japan, China, and Mexico. With an unwavering commitment to enhancing patient outcomes and procedural efficiency, Retriever Medical remains at the forefront of driving progress and excellence in healthcare.
Retriever Medical’s trademarks include Retriever Medical the Retriever Medical logo, ClotHound, ClotHound Blue, ClotHound Gold, ClotHound ACE, Clear ACE, ACE, VORS and Blood Genie. DogCurve, DogLeg, and DogTail are registered trademarks.
For more information, please visit www.rtvmed.com.
Investors Contact:
Ben Bobo
Phone: 714.654.2367
Email: [email protected]
Safe Harbor Statement
This press release includes statements that look forward in time or that express management’s beliefs, expectations or hopes. Such statements are forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. These statements include, but are not limited to, the anticipated approval of pending and future patent applications related to Retriever Medical’s ClotHound, ClotHound ACE, Clear ACE, VORS and Blood Genie Technology, the Company’s future patent application filings and the protection of the Company’s intellectual property provided by any patents that issue. These statements are based on current information and belief and are not guarantees of future performance. Among the risks and uncertainties that could cause actual results to differ materially from those indicated by such forward-looking statements include that pending and future patent applications related to Retriever Medical’s ClotHound, ClotHound ACE and Blood Genie Technology may not result in issued patent, that the issuance of any patents may be delayed, that the allowed claims, if any, may not be in line with the Company’s expectations, that the Company may not be successful in enforcing its patents, and the risk factors detailed from time to time in the Company’s periodic Securities and Exchange Commission filings. By making these forward-looking statements, the Company does not undertake to update them in any manner except as may be required by the Company’s disclosure obligations in filings it makes with the Securities and Exchange Commission under the federal securities laws.
Ben Bobo
Retriever Medical, Inc.
email us here
Visit us on social media:
Twitter
LinkedIn
![]()
Originally published at https://www.einpresswire.com/article/698973703/retriever-medical-advances-innovation-with-filing-of-u-s-patent-application-for-vascular-occlusion-removal-system


